New Tool Monitors Treatment Response to Cancer Relapse
Brain and other central nervous system (CNS) cancers are the leading cause of childhood cancer deaths in the United States. This type of cancer is the second most common type of childhood cancer after leukemia.
Physicians treating CNS tumors with surgery, chemotherapy, radiation, or a combination of these therapies have relied on a series of magnetic resonance imaging (MRI) scans to monitor whether treatment is working or not. But MRI scans can’t detect microscopic disease that could indicate whether cancer cells remain or have returned.
To fill this gap, researchers have been looking for reliable, tumor-specific biomarkers. They know from previous studies in adults that primary tumors shed circulating tumor cells (CTCs) into a patient’s bloodstream and that CTCs could be reliable biomarkers for CNS tumors.
Pediatric researchers at the University of Texas M.D. Anderson Cancer Center wanted to know whether a liquid biopsy tool that relies on detecting vimentin, a structural protein on the surface of many cancer cells, would work to capture and isolate CTCs in blood samples taken from children with CNS tumors.
The researchers who published their study in Cancers also wanted to know whether automating the CTC capture method would improve their previously validated manual method.
How they captured and identified tumor cells
The researchers’ liquid biopsy approach captures cells with cell-surface vimentin (CSV) to isolate CTCs from patients’ blood, which can provide information about their cancer and monitor their ongoing treatment.
The study authors had previously found that the manual liquid biopsy approach detected CTCs in adults with different types of cancer. In the current study, the researchers wanted to automate their method to boost its sensitivity and to capture CTCs from CNS tumors.
The researchers enrolled 62 participants in their study: 58 children (median age 13 years) who were diagnosed with CNS tumors and four healthy adolescents (median age 16 years) who made up the comparison group. Forty-five of the participants with cancer had malignant tumors, including seven whose cancer had metastasized.
The researchers took blood samples from all the participants to isolate and capture the tumor cells. After removing denser cells unlikely to contain CTCs, the researchers loaded the samples onto a machine that is equipped with a microchip. This microchip is coated with an antibody that recognizes CSV, which causes the CTCs to attach to it, but allows other types of cells to flow away. The cells on the chip were then stained so that they could be counted and identified.
Did this automated tool work?
The automated method successfully captured CTCs in 50 of the 58 pediatric patients (86%). There were no significant differences in CTC detection based on patient characteristics, such as gender, age, disease status, or type of cancer therapy.
Overall, the automated CSV-CTC capture tool was highly accurate in identifying patients with and without CNS tumor cells (meaning that the test was both sensitive and specific). The tool was also highly accurate in predicting the presence of CNS tumor cells, but it only predicted their absence about one-third of the time.
“This is the first study to demonstrate the detection of CTCs using CSV as a biomarker in pediatric CNS tumors, including ones that are malignant and have metastasized,” said Shulin Li, Ph.D., principal investigator of this study and professor of pediatrics at UT Anderson Cancer Center.
Also, compared to the researchers’ previous manual CTC capture and identification process, the automated CTC isolation process increased the sensitivity of CTC detection rates by about 10% and decreased sample processing times.
“This study also showed the value of automating the CSV-CTC capture process with a microchip. This tool could make it easier to monitor patients’ response to treatment and identify cancer relapse earlier,” said Shawn Mulvaney, Ph.D., a health science administrator in the NIBIB Division of Applied Science & Technology (Bioimaging).
The research team also wanted to show that they could identify a specific mutation that has been associated with worse prognosis among patients with midline gliomas. When they analyzed the CTCs captured from patients with these types of tumors, they were able to detect this mutation among 75% of the samples.
Next steps
The main study limitations were the small sample size and the fact that the microchip technology required additional steps to confirm the identification of CTCs. While larger studies are needed to validate the findings in this study, the detection of CTCs in patients with CNS tumors can potentially be used to confirm the diagnosis of inoperable, difficult-to-biopsy tumors such as brainstem gliomas and optic pathway gliomas.
“Our tool also has the potential to help physicians know early on which patients will respond to treatment. When they know a tumor isn’t responding, they could enroll the patient in a clinical trial with innovative therapy,” said Wafik Zaky, M.D., lead author and associate professor of neuro-oncology at the University of Texas M.D. Anderson Cancer Center.
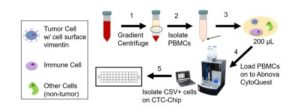
Researchers’ workflow of how they first isolated peripheral blood mononuclear cells (PBMCs) from blood samples, and then isolated the circulating tumor cells (CTCs) with cell surface vimentin (CSV). Credit: Long Dao, Ph.D., a former postdoctoral fellow at the University of Texas M.D. Anderson Cancer Center.
Source